-
Comparing Graphene and Graphyne
Jan 31, 2020 | ACS MATERIAL LLCWhat's the difference between Graphene and Graphyne?
Impossibly tiny, unseen particles that can be used for the applications described below as well as many other things may seem like something out of futuristic science fiction stories, but, in fact, a new class of materials is reshaping the world as we know it today. Nanomaterials, defined as products no larger than 100 nanometers thick (for comparison, a human hair is roughly 80,000 nanometers thick), are responsible for a relentless coalition of innovations that promise to solve many of the world’s most vexing problems. Here are some of the extraordinary applications of both graphene and graphyne:
- Durable, water- and stain-resistant textiles for high fashion and sporting equipment
- Strong, lightweight construction and automotive materials
- Safe, comfortable cosmetic UV filters
- Efficient recycling of greenhouse gases into clean fuel
- Early disease detection, close disease monitoring, and advanced biosensors for disease prevention
- Lightweight, efficient energy storage
- Flexible, translucent, conductive screens
- Nanoscale electronic components that conduct electricity without creating heat or friction
While the idea of nanomaterials has been floating around for a while, the real wave started in 2004 with a singular discovery: graphene. Researchers discovered they could peel off individual layers from a graphite pencil until only one layer remained: a thin sheet of carbon atoms, one atom thick, tightly joined together in a honeycomb lattice. This 2-dimensional, transparent, almost weightless material, conducts heat and electricity, is stronger than steel, more flexible than a rubber band, and is impermeable to gases.
Since then, more than a dozen 2-D materials have been developed and theorized. One of these promising nanomaterials is graphyne, a close relative of graphene. Both graphene and graphyne are 2-D allotropes of carbon that hold exciting promise in a variety of fields, but subtle differences in their structure make a significant impact in how they function.
The Bonds Make All the Difference
Both graphene and graphyne are single-atom thick sheets of carbon atoms. In graphene, individual carbon atoms bond together in a strict hexagonal pattern. Graphene bonds drift between single and double bonds; the result is bonds that are so strong and so tight that the lattice is virtually without defect.
Graphyne bonds look a bit different. Single sheets of graphyne consist of carbon atoms joined together in varying combinations of double and triple bonds; the different combinations create different structures that all potentially have different properties (Graphyne-n). While there’s only one graphene, there may be a vast number of types of graphynes, each with its carbon atoms bonded in subtly different arrangements.
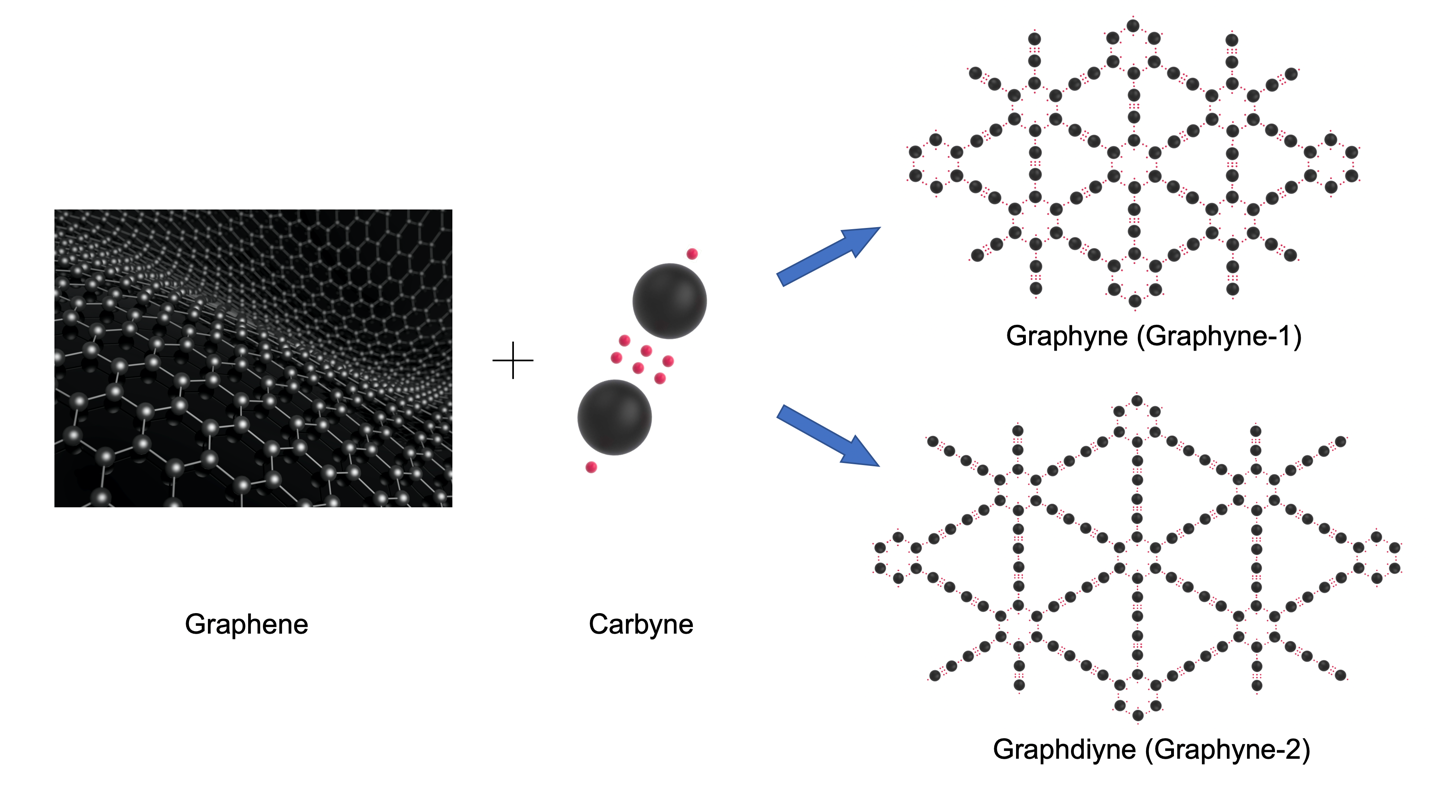
While the bonding pattern of the carbon atoms in graphene and graphyne constitute an important difference between the two materials, the other significant difference at this moment is that graphene physically exists and graphyne is only theoretical. Graphene is used in research labs and industries and it’s been tested in a number of different applications; you can even purchase graphene on the internet from advanced material suppliers like ACS Material. On the other hand, graphyne exists only in theoretical computer models and only graphdiyne can be produced in small samples. Nevertheless, it’s possible to theorize how these materials might function differently from one another.
Electrical Conductivity
One of the features of graphene that makes it so exciting is its tremendous ability to conduct electricity. Because graphene’s bonded carbon atoms each have one loose electron that floats above the surface of the sheet, its electronic energy levels create shapes called Dirac cones that allow electrons to travel in any direction as if they have no mass similar to particles of light; in technical terms, graphene conducts electricity extremely fast.
Graphene’s greatest strength is also a potential weakness; with graphene, electricity flows in multiple directions extremely rapidly, making it impossible to control the flow in only one direction or even to shut off the flow. This is where graphyne comes in; one graphyne configuration, 6,6,12-graphyne, has a structure that also creates Dirac cones over the double and triple bonds; however, in this configuration the Dirac cones aren’t perfectly conical. A vertical cross section of the cones on the short side of the rectangle is an inverted triangle, but a vertical cross section in the other direction is curved, looking more like a parabola. This distortion should result in conductance that depends on the direction of the current. In other words, 6,6,12-graphyne may allow electrons to flow one way in a defined direction at the same speeds found with directionally independent graphene, making it more suitable than graphene for many electronic applications.
Additional Differences
It’s possible to theorize a number of other differences between these two sister materials and the implications for possible applications.
- Strength: The triple bonds in graphyne would likely make this material more rigid than graphene. As a result of increased rigidity, graphyne may be less flexible, more brittle, and less mechanically strong overall than graphene.
- Porosity: Graphyne has nanopores between atoms that are large enough for molecules the size of water to pass through, whereas the structure of graphene is very tight and generally impermeable. As such, graphyne may be useful for such applications as desalination; the nanopores are large enough for water to pass through, but not salt.
- Band Gap: Early indications are that the band gap of graphyne is mechanically tunable, which facilitates the manufacture of a variety of transistors, including those that depend on a specific on-off ratio.
The Graphyne Series from ACS Material
The exact differences between these two materials won’t be known until a sheet of graphyne is actually created, but many of the building blocks are already in place. One of these is graphdiyne, a variant of graphyne. Graphdiyne contains only two acetylene linkages in each unit and its carbon chains are double the length of the carbon chains in graphyne. As a result, graphdiyne is softer and much less rigid than graphyne. However, it’s an excellent semiconductor in theory and in practice.
ACS Material’s graphyne series includes graphdiyne powder and graphdiyne monomer HEB-TMS. Both of these advanced nanomaterials show promise for helium chemical and isotopic separation, water filtration and purification, and in developing faster transistors and nanoscale electronic devices.
ACS Material is your primary source for industry-leading innovative, high-quality nanomaterials. For prices and specific product details and specifications, please see our website or contact our team.