-
Si/C Composites for Battery Materials
Nov 06, 2018 | ACS MATERIAL LLCLithium-ion batteries (LIB’s) are well-suited for fully electric and hybrid electric vehicles due to their high specific energy density in comparison to other rechargeable cell options; however, their suitability depends on the quality of the anode material within.
Silicon Carbide (Si/C) composites are a semiconductive material where silicon is highly dispersed within a carbon matrix. Si/C composites exhibit not only acceptable faradaic yield at the first cycle, but also large capacity and good rechargeability. These are essential and highly desirable properties, making Si/C composites worth considering for use as anode material within lithium-ion batteries.
Introduction
Lithium-ion batteries have been widely used as power sources in all types of modern portable equipment. They show great promise for electric transportation systems, including electric vehicles (EVs) and hybrid electric vehicles (HEVs).1-3
So far, many kinds of anode material for these Lithium-ion batteries have been investigated, such as graphitic carbons,4 disordered carbons,5 tin-based materials, nitrides, phosphides, and oxides. However, only graphitic carbons are commercially available today due to their excellent charge and discharge cycling behavior.
That being noted, however, the theoretical Li-storage capacity of graphite is limited to 372 mAh/g.6 Most recently, there has been considerable interest in developing silicon anode material for lithium-ion batteries due to its high theoretical capacity of 4200 mAh/g and low electrochemical potential versus Li/Li+.7-8
One drawback associated with silicon battery anode material is the very large volume change during the alloying/dealloying process, potentially resulting in pulverization of the electrode materials and poor cycle performance.9 To overcome this problem, tremendous efforts have been made to investigate Si/C material for battery anodes since 1999.10 The mechanical properties of graphitic or amorphous carbon enable it to buffer the volume expansion of Si, while the high conductivity of C can efficiently complement the high lithiation capacity of Si.
Synthesis
When graphite or amorphous carbons are used as matrices, Si/C composites show some promising properties, such as high specific capacity and good cycling stability. Carbon plays an important role in electrode reliability because of its small volumetric change, high electronic conductivity, and its ability to limit volume expansion of Si during electrochemical cycling.
The preparation of Si/C composites begins with first dissolving Polyvinyl chloride (PVC) in 1,2-epoxypropane. Nanosized silicon (<100nm) and fine graphite (1-2 µm) powders are added into the PVC solution and homogeneously mixed under ultrasonic action. When petroleum pitch is used as the precursor, a tetrahydrofuran solvent is used. The solvent is evaporated while stirring to get a solid blend. The blend precursor is then gradually heated to 900°C. After pyrolysis at 900°C for 2 hours, the furnace is cooled. The resulting Si/C composite samples are ground and sifted. The composition of the composites is estimated on the basis of no weight loss of graphite and silicon during pyrolysis.
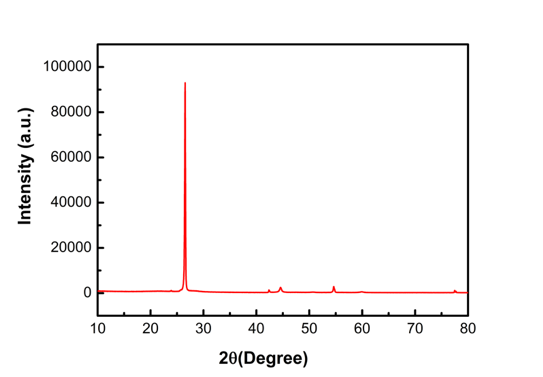
There are two types of Si/C composites available on our ACS Material online store: Type A with a Si content of 8wt% and Type B with 18wt% Si content. The XRD pattern for our Type B Si/C Composite Anode Material can be seen below in Figure 2. From the chart, one can see peaks at 28.5°, 47.5°, 56.2°, and 76.5°, which can be attributed to crystalline reflections of the Si. From this, one can conclude that there is no phase of inert silicon carbide (SiC) observed for the Si/C Composite.
Applications of Silicon Carbon Batteries
The main challenge in seeking the perfect anode materials to be used within lithium-ion batteries is the strong volume effect, which is the main reason for the poor cycling performance of the silicon insertion host.
The small host particle size can enhance dimensional and cycling stability. Therefore, we have attempted to disperse and embed nanosized silicon particles in a carbon matrix with low volume expansion for lithium insertion. By this method, the total volume change of the electrode material can be controlled at a rational level.
Electrode fabrication and electrochemical testing for Si/C composites as a negative electrode material for LIBs are described as follows. To prepare Si/C composite powders for the electrode material, 86% active material, 6% carbon black, and 8% polyvinylidene fluoride (PVdF) binder in n-methylpyrrolidone (NMP) are homogeneously mixed under ultrasonic action. The resulting slurry is coated on Ni foam (ɸ1.25 cm). After the solvent evaporates, the electrode is dried under vacuum at 120°C and finally pressed with 4 MPa.
Electrochemical properties of the active materials are evaluated via coin cells containing 1 M LiPF6/ethylene carbonate (EC)+ dimethyl carbonate(DMC) (1:1 in volume) electrolyte, and a lithium sheet acts as the counter electrode. The charge and discharge current for testing is set to 125 mA/g, and the voltage cut-off is controlled between 0.02 and 1.5 V. It is worth noting that the electrochemical and cycling performance of a particular Si/C composite also depends on the design and assembly of the final lithium-ion battery.
Consumer Electronics
Manufacturers use silicon-carbon batteries to enhance battery capacity without increasing size and to improve efficiency in consumer electronics. The controlled-volume expansion of the Si/C anode enables batteries to operate reliably and maintain performance without rapid capacity loss. Thus, the battery lasts longer per charge and shows slower performance degradation over time. Such a lithium cell supports faster charging, which is essential for modern portable devices.
Electric Vehicles
The silicon-carbon batteries play a major role in increasing driving range and improving battery durability. Moreover, the Si/C anode structure minimizes mechanical damage, thereby improving cycle life under high-energy, high-density charging conditions. So, this allows EV batteries to provide more energy while ensuring stability across thousands of charge-discharge cycles.
Energy Storage Systems
Batteries need to operate reliably for extended periods in motionless energy storage systems to ensure long-term, effective performance. Manufacturers use silicon-carbon batteries because their controlled volume change minimizes internal damage over time. Also, these Si/C anodes improve long-term cycling stability and make them suitable for daily charge-discharge applications, such as renewable energy storage.
Fast Charging Devices and Power Tools
To handle the high current without structural failure, fast-charging devices need high-performance batteries. As a result, manufacturers use silicon-carbon batteries because the carbon matrix maintains electrical contact, while silicon stores more lithium. Thus, silicon anode reduces internal stress during fast charging, helping the battery maintain consistent performance during repeated cycling.
Medical and Wearable Devices
A Si/C battery is essential for wearable devices in health care centers and hospitals, providing stable performance while remaining compact. Besides, silicon-carbon battery technology is lightweight and offers higher energy density, enabling longer runtime between charges. The improved cycling stability of lightweight silicon anode batteries enables consistent operation without risk or compromise to device health.
Advanced Aerospace Systems
In aerospace, we need lightweight, high-strength battery materials to improve reliability and performance. Manufacturers use silicon anode batteries to store maximum energy in a compact battery mass. Although conventional lithium-ion batteries are better than older lead-acid options, Si/C batteries are lightweight and next-generation alternatives.
Sensors And Industrial Electronics
Industrial sensors and monitoring systems mostly operate in continuous cycles to ensure safety and performance. In this regard, manufacturers use Si/C batteries to maintain the structural integrity and capacity for long operational periods.
In industries, sensor systems monitor temperature, gas levels, and equipment health. The silicon-carbon anode design limits mechanical damage from repeated charging and discharging cycles during device operation. Beyond this, the improved resistance to cycling degradation ensures that the sensor performance remains stable over time. Silicon battery technology is well-suited for industrial environments where minimal severity, longevity, and reliability are critical demands.
Ending Up Discussion
Si is a promising negative electrode material for boosting the high energy density of LIBs because of its high specific capacity. However, significant challenges must be overcome before Si negative electrodes can be used in practical LIBs. To overcome potential drawbacks during lithium insertion/extraction, designing an appropriate Si/C composite as a negative electrode material for LIBs is key.
Although much progress has been made regarding the study of Si/C composites as a negative electrode material, there are still many topics left unstudied in the field. Further research on lithium-ion batteries is needed to improve the initial coulomb efficiency of LIBs based on Si/C negative electrodes because it will be critical for practical applications.
Currently, it is quite expensive to prepare Si/C nano composites by the methods known today. This means it is necessary for us to continue working on developing new methods with low cost and high possibility for upscaling.
FAQ
Q. What Are the Benefits of Sic Batteries?
A. SiC batteries offer countless benefits, including higher energy density, improved efficiency, and reduced energy loss as heat. Silicone’s higher energy density enables the packing of more energy into a smaller volume, resulting in more compact devices.
Q. Are Silicon-Carbon Batteries Better?
A. Yes, silicon-carbon batteries are far better than the conventional graphite-anode lithium-ion batteries. They have higher energy density, improved device performance, and faster charging. Additionally, they can even operate seamlessly in cold temperatures, which is a significant improvement over other battery types.
Q. Do Silicon Carbide Batteries Last Longer?
A. Yes, Sic batteries last longer than traditional lithium batteries. It reduced degradation and improved stability.
Q. Does Tesla Use Silicon Carbide Batteries?
A. No, Tesla doesn’t use silicon carbide in battery cells. However, it utilizes silicone carbide chips in its power electronics to improve the device's performance.
Q. Can Silicon-Carbon Batteries Be Used in Cars?
A. Yes, silicone-carbon batteries can be used in electric vehicles due to their exceptional qualities. They provide high energy density, safer charging, and a longer lifespan.
Q. How Fast Can You Charge a Silicon-Carbon Battery?
A. Silicone-carbon batteries can charge very quickly; some smartphones can show an 80% charge in approximately 15 minutes. While for electric vehicles, these batteries promise as little as 10 minutes of charge to gain 400 miles of range.
Q. What Are the Limitations of Silicon Carbide Batteries?
A. High production cost, material expansion, and complex manufacturing are the significant limitations for silicone carbide batteries.
Q. What Are the Three Types of Batteries?
A. The first one is primary batteries that are non-rechargeable, like alkaline. The second most famous type is secondary batteries that are rechargeable, such as lithium-ion and lead-acid batteries. Lastly, the flow or reserve batteries are preferred for the special niche-based use.
References
1. Scrosati B, Hassoun J, Sun Y K. Lithium-ion batteries. A look into the future. Energy & Environmental Science, 2011, 4 (9): 3287-3295.
2. Hua W, Wang Y, Zhong Y, et al. An Approach towards Synthesis of Nanoarchitectured LiNi 1/3 Co 1/3 Mn 1/3 O2, Cathode Material for Lithium Ion Batteries. Chinese Journal of Chemistry, 2015, 33 (2): 261-267.
3. Dunn B, Tarascon J M. Electrical energy storage for the grid: a battery of choices. Science, 2011, 334 (6058): 928-935.
4. Mohri M, Yanagisawa N, Tajima Y, et al. Rechargeable lithium battery based on pyrolytic carbon as a negative electrode. Journal of Power Sources, 1989, 26 (3-4): 545-551.
5. Sato K, Noguchi M, Demachi A, et al. A Mechanism of Lithium Storage in Disordered Carbons. Science, 1994, 264 (5158): 556-558.
6. Hossain S, Kim Y K, Saleh Y, et al. Comparative studies of MCMB and C/C composite as anodes for lithium-ion battery systems. Journal of Power Sources, 2003, 114 (2): 264-276.
7. Netz A, Huggins R A. Amorphous silicon formed in situ as negative electrode reactant in lithium cells]. Solid State Ionics, 2004, 175 (1–4): 215-219.
8. Winter M, Besenhard J O, Spahr M E, et al. Insertion Electrode Materials for Rechargeable Lithium Batteries. Advanced Materials, 2010, 10 (10): 725-763.
9. Slinkin A A, Loktev M I, Klyachko A L, et al. Study of the ESR method of the nature of the redox centers of mordenites in reactions of formation of cation radicals in the absorption of aromatic hydrocarbons. Bulletin of the Academy of Sciences of the Ussr Division of Chemical Science, 1975, 24 (5): 936-941.
10. Xu C, Guo Y, Xiao Q, et al. Synthesis and characterization of large, pure mordenite crystals. Journal of Porous Materials, 2012, 19 (5): 847-852.
11. Yang J, Takeda Y, Imanishi N, et al. Ultrafine Sn and SnSb0.14 powders for lithium storage matrices in lithium-ion batteries. Journal of the ElectrochemicalSociety, 1999, 146(146):4009-4013.
12. Dimov N, Kugino S, Yoshio M. Carbon-coated silicon as anode material for lithium ion batteries: advantages and limitations. Electrochimica Acta, 2003, 48 (11): 1579-1587.
13. Magasinski A, Dixon P, Hertzberg B, et al. High-performance lithium-ion anodes using a hierarchical bottom-up approach. Nature Materials, 2010, 9(4):353-358.
14. Yang J, Winter M, Besenhard J O. Small particle size multiphase Li-alloy anodes for lithium-ionbatteries. Solid State Ionics, 1996, 90(1-4):281-287.