-
Graphene: Super Strength for the 21st Century
Jan 08, 2020 | ACS MATERIAL LLCThe World of Carbon
Peek deep inside a dark, chalky chunk of graphite and a clear, bright diamond and you’ll discover a startling truth: these radically different materials are, in fact, fundamentally the same–they’re both made exclusively from carbon. The different properties of graphite and diamonds are the result of the way individual carbon atoms bond together.
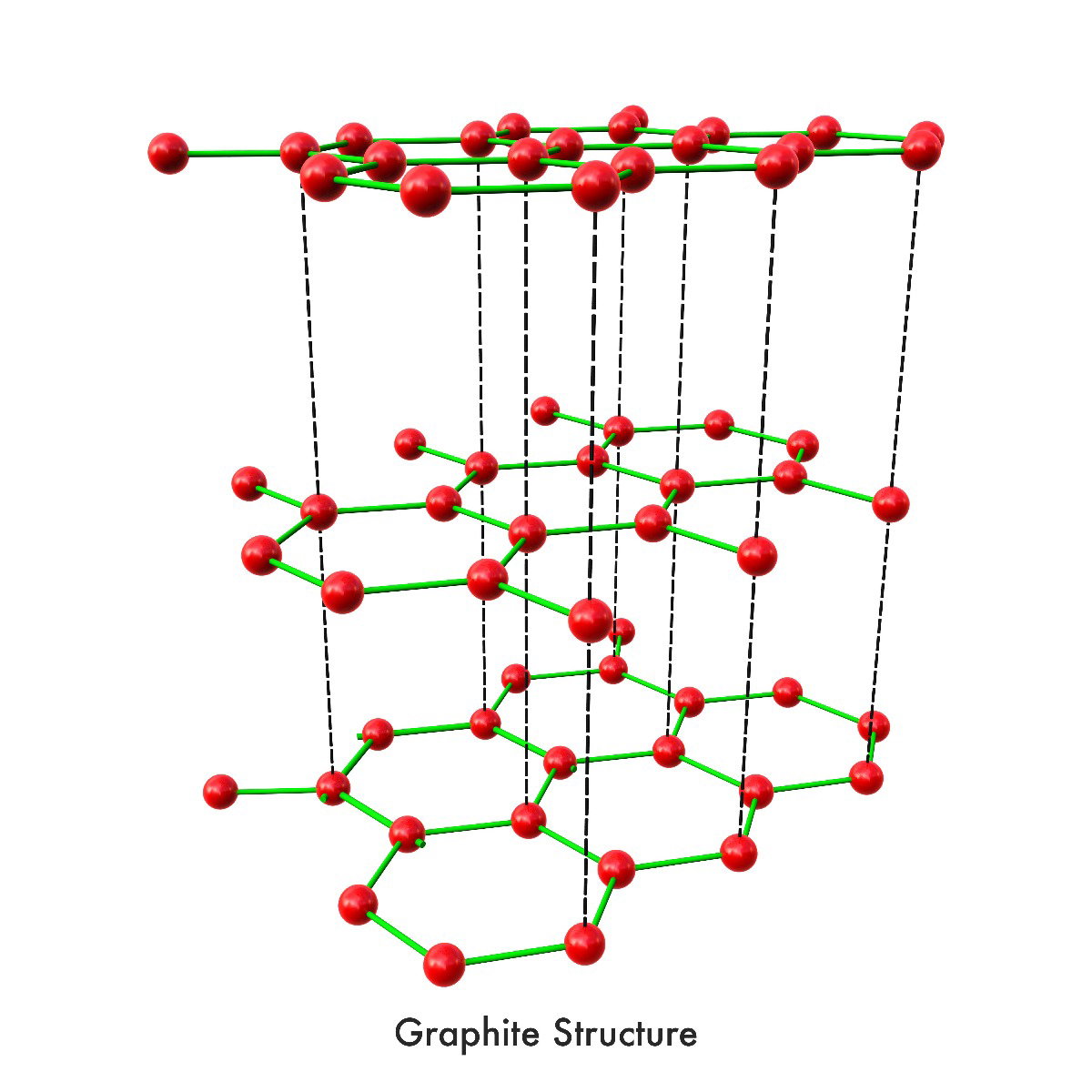
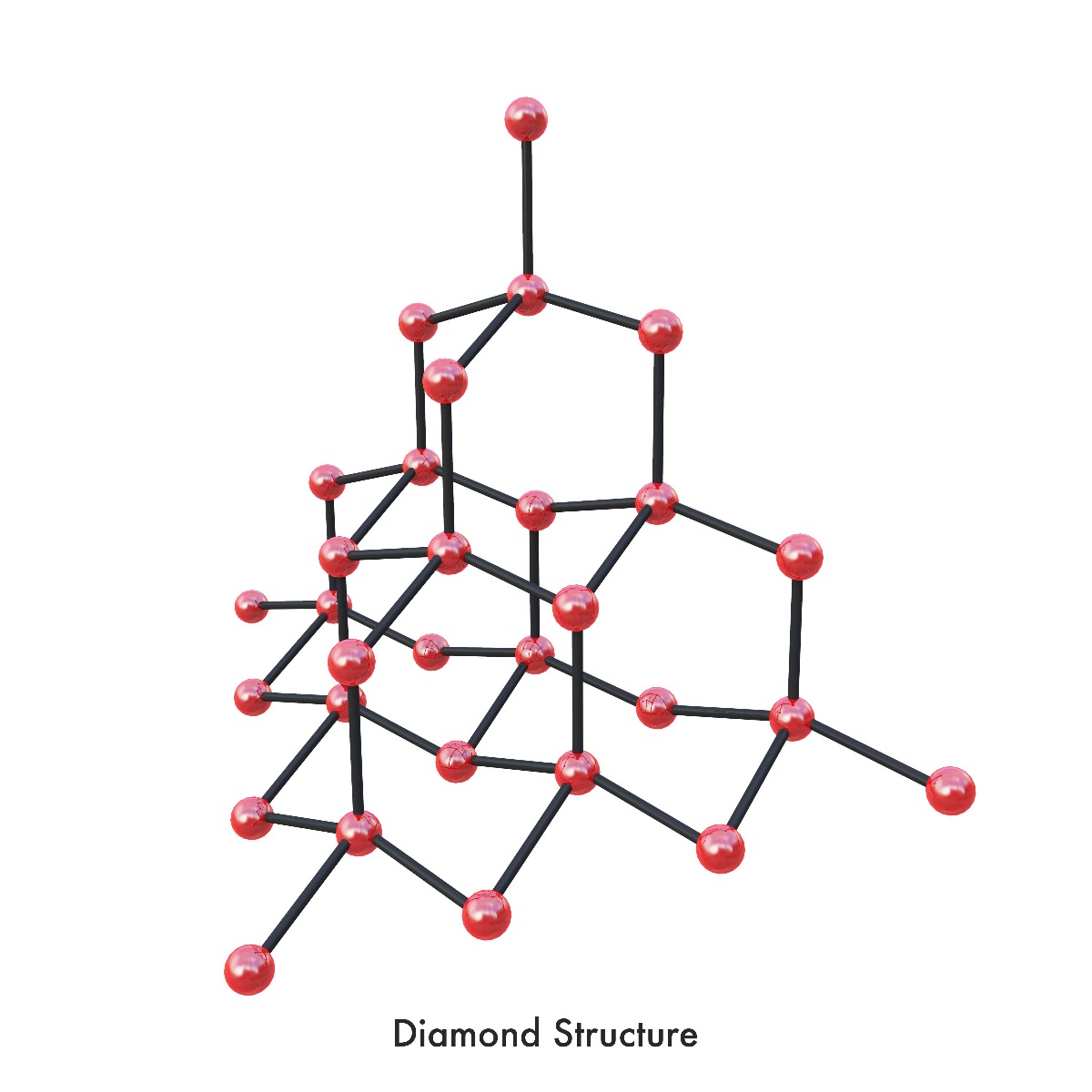
These different bonding patterns are called allotropes of carbon. When carbon atoms bond trigonally to three other carbon atoms in a plane of hexagonal rings and stacks of these planes are held together with van der Waals forces, the result is graphite, a carbon allotrope soft enough to leave behind streaks when pulled across paper. Conversely, when carbon atoms bond together under high pressure in tetrahedral bonds that form three-dimensional 6-sided rings, the diamond allotrope is formed, creating the hardest material currently known. Carbon allotropes are an endlessly fascinating topic; new allotropes are being discovered each year with wildly different properties, opening up new possibilities for understanding and innovation. According to Samara Carbon Allotrope Database, 522 unique 3-periodic carbon allotropes corresponding to 494 topological types have currently been discovered. One of these many carbon allotropes is graphene.
a)
 b)
b)a) Graphite (strong covalent bonded hexagonal rings and inter-layer bonded stacked planes); b) Diamond (strong tetrahedral covalent bonded 3D 6-sided rings)
A Material with Unbelievable Strength
Graphene is, very simply, a single “sheet” of graphite. Remember that graphite is a stack of planes of hexagonal rings of carbon atoms; one isolated plane, or sheet, of hexagonal carbon rings is an allotrope called graphene. Graphene has been around in theory for more than a hundred years, but it wasn’t until 2004 that this material really took off. In 2004, researchers Andre Geim and Kostya Novoselov at The University of Manchester separated single-atom-thick graphene layers from graphite and transferred them onto a thin SiO wafer using the scotch-tape exfoliation method. Since then, research into graphene and its properties has exploded across all fields and scientists and innovators around the world are working tirelessly to develop methods to produce graphene in bulk quantities.
But why is everyone so excited about graphene? Why is this very simple carbon allotrope attracting so much attention? The answer: graphene is getting all the buzz because graphene is amazing. Graphene is lightweight, virtually transparent, and demonstrates extraordinary electrical and mechanical properties. Graphene is also super strong, 100 times stronger than steel! This unbelievable strength, placed among its other amazing properties, makes graphene a truly exciting material.
Graphene: The Superhero of Carbon Allotropes
Measurements of the intrinsic strength of graphene consistently show that it is the strongest material ever discovered. Graphene has a simple structure: carbon atoms bonded side by side in a hexagonal pattern in a sheet one atom thick. This simple configuration plus the enormous strength of the individual bonds results in very few structural defects across the surface. Put another way, there are virtually no weak links in a graphene chain of superstrong carbon links. The result is a material with an intrinsic tensile strength of 130 GPa and a Young’s modulus of 1 TPa. By comparison, steel has a tensile strength of 0.4 GPa. Graphene has a breaking strength of 42 N/m and steel has a breaking strength of 0.25-1.2x109 N/m2. Using this measure, a hypothetical steel film the same thickness as graphene would give a breaking strength of 0.084-0.40 N/m, making graphene 100 times stronger than steel. When Geim and Novoselov’s 2010 Nobel Prize in Physics was announced, the incredible strength of graphene was illustrated by saying that a hammock made from 1 square meter of graphene would support a 4 kg cat but would weigh only as much as one cat whisker.
Exploring the Possibilities
Graphene’s strength in combination with its other electrical and mechanical properties open the door for many exciting applications. ACS Material has a range of high-quality graphene products available for researchers around the world studying and experimenting with this exceptional material. To learn more about graphene and its applications, visit the ACS Material website today.
Related ACS Material Products