-
GlowDots
May 29, 2018 | ACS MATERIAL LLCFacile synthesis of multi-colored, protein-based, metal-free, stable, nontoxic and pH sensitive advanced functional nanoparticles (GlowDots) is now a reality. Controlled cross-linking of bovine serum albumin has resulted in the formation of spherical nanoparticles of 35 nm in diameter with a size distribution of ±10 nm. The nanoparticles are then conjugated with specific dyes to produce white-emitting particles with tunable excitation wavelengths. These nanoparticles are a chemical novelty because their particle size, size distribution, stability, surface chemistry and emission properties are under full chemical control and their size and absorption/emission properties can be independently tuned. Up to 100 dye molecules were attached to each particle on average and as a result, the particles acquired strong absorption cross-sections as well as high brightness. The white fluorescence of GlowDots is strongly sensitive to pH over a range of pH 2–11 and pH-induced emission changes are fully reversible. The particles readily entered HeLa cells and the emission color depended on particle location in the live cells, which is most likely due to the environment directly surrounding the particles. These are the very first reports of multi-colored and white-emitting advanced functional nanoparticles that are biodegradable, sensitive to pH and amenable for live cell imaging.
Introduction
Nanoparticle-based pH sensors are currently being developed for biological and chemical applications,[1] but they have certain drawbacks such as toxicity, limited pH range, difficult synthesis or poor compatibility for biological applications.[2–8] This report introduces a biocompatible and modular synthesis of pH-sensing, fluorescent, metal-free, white-emitting and advanced functional nanoparticles derived from a common and inexpensive protein, bovine serum albumin (BSA). These GlowDots can be used as alternatives to quantum dots. The use of a protein matrix for the particle synthesis renders them biodegradable at pre-programmed rates and the presence of a variety of multiple functional groups on the protein surface are convenient for attaching fluorescent dyes, drugs, or ligands to the particles.
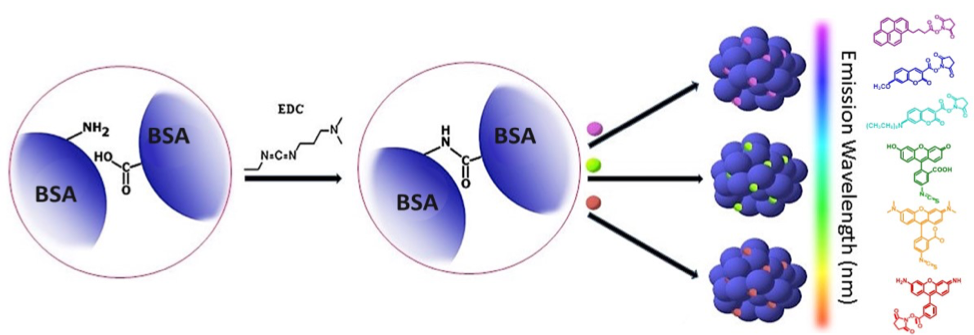
Scheme 1: Synthesis of multi-colored GlowDots (340, 350, 432, 494, 543 and 576). BSA is crosslinked using carbodiimide chemistry, followed by conjugation of specific dyes to obtain color. White emitting particles (254W, 365W, and 402W) have the same procedure except red, green and blue dyes are conjugated to one particle in a particular proportion to generate white emission.
The choice of BSA for advanced nanoparticle synthesis is justified by multiple reasons. BSA has 59 amino groups from lysines and 99 COOH groups from aspartate and glutamate side chains which are convenient for self-crosslinking of the protein to form nanoscale particles under complete chemical control.[9] BSA binds a number of small molecules, metal ions and signal peptides, therefore, particles derived from BSA can potentially serve as carriers for these and other ligands.[10,11] BSA-derived particles also have a high potential for controlled biodegradation by proteases at pre-programmed rates when released into the environment.[12,13] Fluorescent-dye labeled BSA particles can be versatile for sensing and the presence of a large number of functional groups with different pKa values on the particles provides an excellent pH-sensing platform, as demonstrated here.[14,15] The particle surface can be readily modified with natural ligands to enhance cellular uptake.
BSA also serves as a model protein for protein transduction into cells, where the protein is directly imported into the cell to trigger specific biological responses. Protein transduction is thought to be less sensitive to hydrolysis by cellular environment than the DNA/RNA transfection approach.[16–19] Certain cells have specific receptors for BSA and can promote receptor-mediated endocytosis of the particles. Hence, advanced functional nanoparticles derived from BSA (nBSA) are expected to be versatile, biocompatible and also scientifically interesting for pH sensing and cellular imaging.
A.
 B.
B. 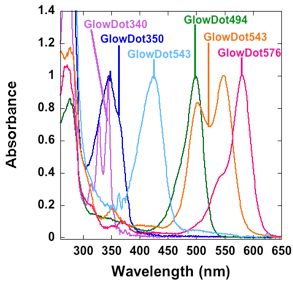 C.
C. 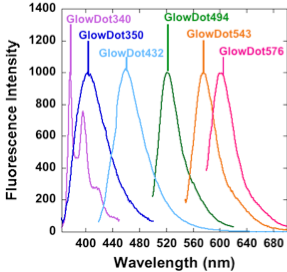
Figure 1: A. Agarose gel of the GlowDots labeled with specific dyes. B. Absorbance spectra. C. Fluorescence Spectra. Particles emit violet, indigo, blue green, orange and red light as demonstrated here, for different practical uses.
GlowDots are synthesized by chemical cross-linking under controlled conditions. The particles are labeled with particular dyes to tune their absorption and emission properties. The samples are purified and assayed on agarose gel (Figure 1A) which indicates their high purity and the absence of the free dye. The absorption and emission spectra of the samples indicates bands that correspond to the respective dyes used for the labeling (Figure 1B, C). The physical parameters of the samples are collected in Table 1.
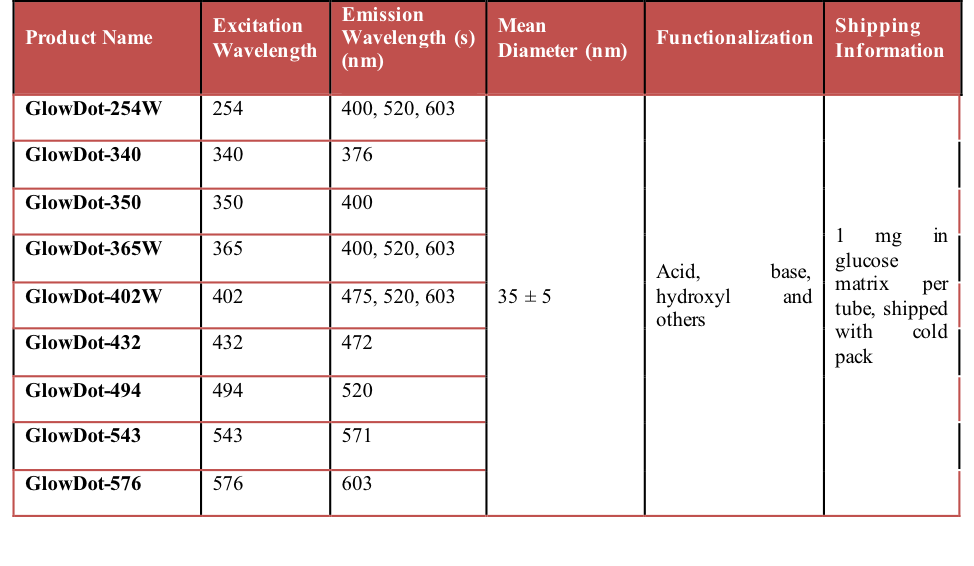
Table 1: Physical parameters of various GlowDot samples.
The chemical novelty of the current approach is the unique ability to independently tune the particle size, color, extinction coefficients, spectral maxima and quantum yields, while keeping the synthetic methodology modular, simple and efficient. Furthermore, the method can be executed in a resource-limited setting. These are significant advantages over other nanoparticles such as quantum dots (QDs). The size and color of QDs cannot be independently tuned, which is a non-issue with GlowDots. Special modifications are required to make QDs water soluble and they are potentially toxic to biological systems.[29] Therefore, we tested GlowDots for specific advantages to complement the utility of QDs in many aspects.
Staining of the HN12 cells with GlowDot494 followed by washing with a buffer, indicated the uptake and localization of the nanoparticles in various parts of the cell (Figure 2). The uptake was complete in less than 3 hours and subsequently the particles were ejected from the cell, which is intriguing.
Another aspect of these particles is that they can emit white light when each particle is labeled with red, green and red fluorophores. White-emitting nanomaterials are of current interest for use in organic light-emitting diodes,[20–23] pollution monitoring,[24] and detection of ions.[25,26] Sensors based on white-emitting particles are expected to be more sensitive to analyses because white light is composed of three color components in specific intensity ratios and any change in one color intensity can produce detectable off-white emission. This is a good strategy for building robust advanced sensors.[27,28] White-emitting particles are now available for biological applications. These can be excited at 254, 365 or 404 nm, depending on the type of particle available. GlowDot365W, for example, emits white light when excited at 365 nm. Sensing with white emitters minimizes interference from background emission and provides greater flexibility in choosing the excitation/emission wavelengths at multiple wavelengths without changing the particles. The presence of multiple pKa groups on a protein particle may contribute to increased sensitivity of the three color intensities to pH, thus providing a simple method to sense local pH.
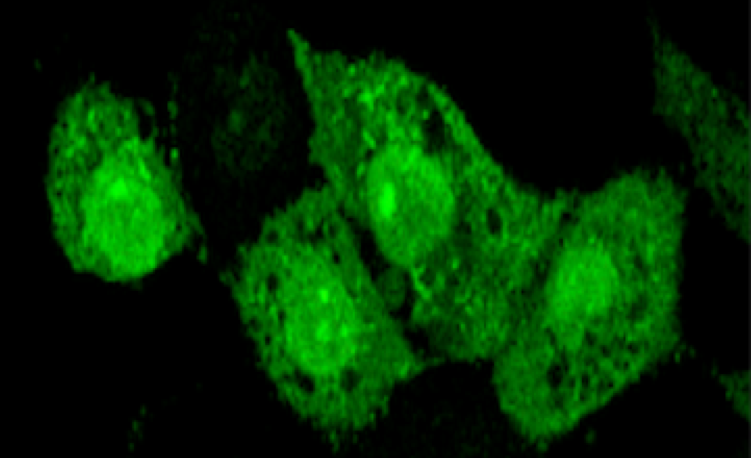
Figure 2. The fluorescence images of HN12 cells stained with GlowDot294 and washed to reveal its presence inside the cells. GlowDot494 can be seen throughout the entire cell as the microscope focuses from the bottom of the cell to the top.
All GlowDot samples were modified with a proprietary ligand on the surface to enhance cellular uptake. After surface modification these cells elicited no toxicity in a variety of cell lines.
Conclusions
Advanced protein fluorescent nanoparticles that emit white light upon excitation at 254 or 405 nm (with chromaticity coordinates close to that of pure white light) as well as multi-colored GlowDots are currently available. Particle formation is under chemical control and the surface chemistry, absorption spectra, and emission characteristics are tuned as desired while keeping the particle size constant, unlike QDs. Due to the large number of copies of the dyes present in each particle, these have very strong absorption and intense emission characteristics that are suitable for imaging applications. The sensitivity of GlowDot emission to pH further enhances their utility to view cellular compartments. These functional protein nanoparticles are white-emitting or multi-colored, biocompatible, cost effective and can be used in applications for sensing.
ACS Material Products:
Graphene Series:
Graphene-like Materials:
Quantum Dots & Upconverting Nanoparticles:
References
1. A. P. Demchenko, Methods Appl. Fluoresc. 2013, 1, 1.
2. A. Burns, P. Sengupta, T. Zedayko, B. Baird, U. Wiesner, Small 2006, 2, 723.
3. J. Liu, M. Yu, C. Zhou, J. Zheng, Mater. Today 2013, 16, 477.
4. M. Ulusoy, R. Jonczyk, J. Walter, S. Springer, A. Lavrentieva, F. Stahl, M. Green, T. Scheper, Bioconjugate Chem. 2016, 27, 414.
5. Z. Li, W. Xu, Y. Wang, B. R. Shah, C. Zhang, Y. Chen, Y. Li, B. Li, Carbohydr. Polym. 2015, 121, 477.
6. S. B. Rizvi, S. Y. Yang, M. Green, M. Keshtgar, A. M. Seifalian, Bioconjugate Chem. 2015, 26, 2384.
7. Z. Wang, A. Bonoiu, M. Samoc, Y. Cui, P. Prasad, Biosens, Bioelectron. 2008, 23, 886.
8. X. Michalet, F. F. Pinaud, L. A. Bentolila, J. M. Tsay, S. Doose, J. J. Li, G. Sundaresan, A. M. Wu, S. S. Gambhir, S. Weiss, Science 2005, 307, 538.
9. K. A. Majorek, P. J. Porebski, A. Dayal, M. D. Zimmerman, K. Jablonska, A. J. Stewart, M. Chruszcz, Mol. Immunol. 2012, 52, 174.
10. A. Buaranaprapuk, C. V. Kumar, S. Jockusch, N. J. Turro, Tetrahedron 2000, 56, 7019.
11. A. O. Elzoghby, W. M. Samy, N. A Elgindy, J. Controlled Release 2012, 157, 168.
12. A. Beloff, C. Anfinsen, J. Biol. Chem. 1948, 176, 863.
13. X. Ma, X. Sun, D. Hargrove, J. Chen, D. Song, Q. Dong, X. Lu, T. Fan, Y. Fu, Y. Lei, Sci. Rep. 2016, 6, 19370.
14. S. Furukawa, H. Shano, T. Mutai, K. Araki, ACS Appl. Mater. Interfaces 2014, 6, 16065.
15. G. Del Frate, F. Belina, G. Mancini, G. Marianetti, P. Minei, A. Pucci, V. Barone, Phys. Chem. Chem. Phys. 2016, 18, 9724.
16. M. Ruponen, S. Arkko, A. Urtti, M. Reinisalo, V. P. Ranta, J. Controlled Release 2009, 136, 226.
17. M. T. Larsen, M. Kuhlmann, M. L. Hvam, K. A. Howard, Mol. Cell. Ther. 2016, 4, 3.
18. R. Treisman, Cell 1985, 42, 889.
19. A. Shyu, M. E. Greenberg, J. G. Belasco, Genes Dev. 1989, 3, 60.
20. E. Park, T. Erdem, V. Ibrahimova, S. Nizamoglu, H. V. Demir, D. Tuncel, ACS Nano 2011, 5, 2483.
21. P. Yang, H. Chen, J. Wang, Q. Che, Q. Ma, Y. Cao, Y. Zhu, RSC Adv. 2014, 4, 20358.
22. H. Tai, C. Cheng, P. Wang, C. Wu, G. Lin, RSC Adv. 2015, 5, 105239.
23. H. Keita, B. Guzelturk, J. Pennakalathil, T. Erdem, H. V. Demir, D. Tuncel, J. Mater. Chem. C. 2015, 3, 10277.
24. S. Bhunia, N. Chatterjee, S. Das, K. D. Saha, A. Bhaumik, ACS Appl. Mater. Interfaces 2014, 6, 22569.
25. X. Lian, B. Yan, Dalton Trans. 2016, 45, 2666.
26. L. Zhi, Z. Wang, J. Liu, W. Liu, H. Zhang, F. Chen, B. Wang, Nanoscale 2015, 7, 11712.
27. J. R. Lakowicz, Principles of Flurescence Spectroscopy, Springer Science+Business Media, LLC, Singapore 2006.
28. H. Sun, K. Almdal, T. L. Andresen, Chem. Commun. 2011, 47, 5268.
29. S. J. Rosenthal, D. W. Wright, Nanobiotechnology Protocols, Humana, Press Inc. Totowa, New Jersey 2005.